Guthrie, K.A. and Yong , M. and Frieze, D. and Corey, L. and Fredricks, D.N. (2010) The Impact of a Change in Antibacterial Prophylaxis from Ceftazidime to Levofloxacin in Allogeneic Hematopoietic Cell Transplantation. Bone Marrow Transplantation, 45 (4). pp. 675-681. ISSN 0268-3369
Preview |
Text (Manuscript)
GuthrieKNatureManuscript081309.pdf Available under License Creative Commons Attribution Non-commercial No Derivatives. Download (91kB) | Preview |
![[thumbnail of Figure 1]](http://authors.fhcrc.org/326/2.hassmallThumbnailVersion/Figure1_fever.jpg) 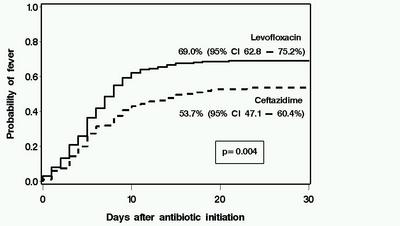 Preview |
Image (Figure 1)
Figure1_fever.jpg Available under License Creative Commons Attribution Non-commercial No Derivatives. Download (41kB) | Preview |
![[thumbnail of Figure 2]](http://authors.fhcrc.org/326/3.hassmallThumbnailVersion/Figure2_bacteremia.jpg) 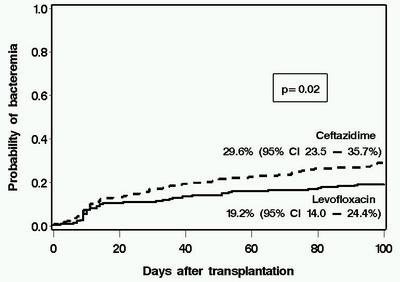 Preview |
Image (Figure 2)
Figure2_bacteremia.jpg Available under License Creative Commons Attribution Non-commercial No Derivatives. Download (41kB) | Preview |
![[thumbnail of Figure 3]](http://authors.fhcrc.org/326/4.hassmallThumbnailVersion/Figure3_any_inf.jpg) 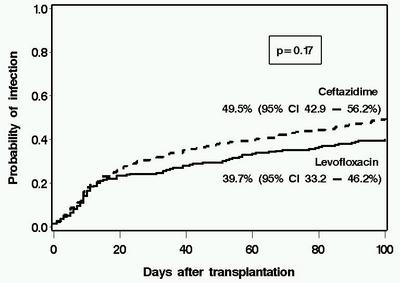 Preview |
Image (Figure 3)
Figure3_any_inf.jpg Available under License Creative Commons Attribution Non-commercial No Derivatives. Download (41kB) | Preview |
![[thumbnail of Figure 4]](http://authors.fhcrc.org/326/5.hassmallThumbnailVersion/Figure4_RFS.jpg) 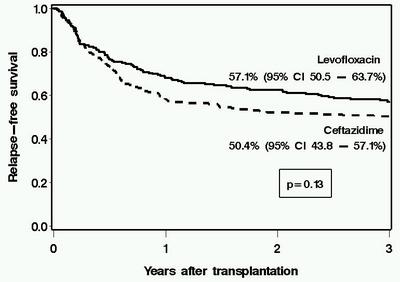 Preview |
Image (Figure 4)
Figure4_RFS.jpg Available under License Creative Commons Attribution Non-commercial No Derivatives. Download (40kB) | Preview |
Abstract
Antibiotic prophylaxis has been used during the initial phases of myeloablative hematopoietic cell transplantation (HCT) for more than two decades. However, the optimal regimen in terms of both cost and clinical effectiveness is unclear. We retrospectively compared the clinical and microbiological impact of a change in antibiotic prophylaxis practice from ceftazidime (n=216 patients with HCT in 2000–2002) to levofloxacin (n=219 patients, August 2002–2005) in patients receiving myeloablative conditioning. Levofloxacin prophylaxis was associated with fever and a change in antibiotics during neutropenia, but this strategy was not associated with any adverse outcomes. Patients receiving levofloxacin had lower rates of significant bacteremia than did those receiving ceftazidime (day 100, 19.2 vs 29.6%, P=0.02). The use of levofloxacin was associated with lower antibiotic acquisition costs. There was no deleterious impact caused by levofloxacin prophylaxis on survival, emergence of antibiotic resistance, detection of Clostridium difficile Ag in stool specimens, incidence of viridans group streptococcal bacteremia or Pseudomonas infections. There was a trend toward lower rates of bacteriuria, wound and bacterial respiratory infections in the levofloxacin than in the ceftazidime group, but these differences were not statistically significant. These data support the use of levofloxacin as prophylaxis in myeloablative allogeneic HCT when prophylaxis is used.
| Item Type: | Article or Abstract |
|---|---|
| Additional Information: | This article is available to subscribers only via the URL above. |
| DOI: | 10.1038/bmt.2009.216 |
| PubMed ID: | 19718062 |
| NIHMSID: | NIHMS138610 |
| PMCID: | PMC2911962 |
| Grant Numbers: | P01 CA018029-31 |
| Keywords or MeSH Headings: | Adult Aged Antibiotic Prophylaxis/methods* Bacteremia/prevention & control* Ceftazidime/therapeutic use* Disease-Free Survival Drug Resistance, Bacterial Female Hematopoietic Stem Cell Transplantation* Humans Male Middle Aged Ofloxacin/therapeutic use* Retrospective Studies Transplantation Conditioning* Young Adult |
| Subjects: | Diseases > Bacterial and fungal diseases Therapeutics > Transplantation > Stem Cell transplantation Therapeutics > Drug Therapy |
| Depositing User: | Library Staff |
| Date Deposited: | 14 Jan 2010 19:05 |
| Last Modified: | 14 Feb 2012 14:43 |
| URI: | http://authors.fhcrc.org/id/eprint/326 |
Repository Administrators Only
 |
View Item |


